Abstract
Objective To evaluate the real-world impact of using a commercially available combinatorial pharmacogenomic (CPGx) test on medication management and clinical outcomes in children and adolescents treated at a tertiary care psychiatry practice.
Methods A retrospective cohort study using our prospectively maintained database of patients undergoing CPGx testing was performed. Only patients with clinical data at the time of ordering CPGx test (pre-baseline), potential medication change visit (baseline) and 8-weeks follow-up (post-baseline) visit were included. Clinical Global Impression (CGI) scores for each visit were calculated. Appropriate statistical analysis, including one-sample t-test, paired t-test and Chi-square test was performed.
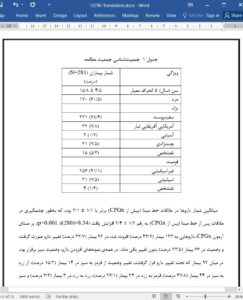
Results Based on the inclusion criteria, 281 (75.9%) of the 370 patients with CPGx testing were included. Their mean age was 15.8 ± 4.5 years (111 females; 39.5%). The average number of medications significantly increased to 2.4 ± 1.2 on the post-baseline visit [t(280) = 8.34, p < 0.001). Medications were added in 123 (43.7%), replaced in 92 (32.7%) patients and remained unchanged in rest. There was no significant association between medication-related adverse effects and psychotropic medication change group (p = 0.27). The study population showed a significant improvement (p < 0.001) in the CGI severity, efficacy, and global improvement indices.
Conclusion In our experience of using CPGx test in a large cohort of children and adolescents during routine clinical practice, three-quarter of them underwent medication change. Additionally, we noted an improvement in clinical outcomes without impacting adverse effects. While the role of clinical judgement in medication changes in our cohort is likely, CPGx may supplement clinical decision making. However, the best use and benefit of CPGx in routine clinical practice needs further investigation.
4. Discussion
We present one of the largest, real-world experience of the use of CPGx test in close to 300 patients seen in child and adolescent psychiatry clinics at a tertiary care center. The study population showed an overall improvement in clinical outcomes after changes in medication based on the results of CPGx testing without a significant increase in adverse effects. This is comparable to a recent meta-analysis of 1556 adults (≥18 years), which concluded that CPGx guided care improves outcomes among patients with major depressive disorder (Brown et al., 2020).