Abstract
CO2 storage in underground saline aquifers is helpful to reduce CO2 emission in the atmosphere, where gas/fluid diffusion and permeation in clay mineral plays a key role in CO2 leakage and underground migration. CO2 Permeability and different fluid diffusivities in clay mineral (montmorillonite, Mt) interlayers are investigated by molecular dynamics (MD). Both CO2 and H2O self-diffusivities increase with water concentration and temperature but show a maximum at the CO2 concentration of 2 molecule/unit-cell unconventionally. The fractional free volume of Mt increases with CO2 concentration but begins to decrease if CO2 concentration exceeds 2, thus giving the reason for the above unusual CO2 self-diffusivity variation. Displacement distribution of CO2 molecules is found to be characterized by logarithmic normal distribution. The mean value of such distribution further supports the self-diffusivity dependence on CO2 concentration. M-S and Fick diffusivities of CO2 are positively related to CO2, H2O concentration and temperature. CO2 permeability is calculated by MD for the first time, which increases with CO2 pressure and H2O concentration but exhibits a turning point at temperature 360 K due to low CO2 solubility at high temperature.
1. Introduction
CO2 storage in underground saline aquifers provides long-term and large-scale storage of CO2, which is a promising way to reduce CO2 emission in the atmosphere. In this process, clay minerals, such as illite, chlorite, kaolinite, and montmorillonite (Mt) (Josh et al., 2012), are the main components of caprocks. Owing to their porous (layered) structure, the clay minerals have remarkable capacity of adsorbing CO2 (Fu et al., 1990; Khosrokhavar et al., 2014). On the other hand, clay mineral has a low permeability and therefore the clay-enriched caprocks show excellent sealing ability to retain injected CO2 (Abdou and Ahmaed, 2010; Gaus, 2010; Gernot et al., 2013). Gas leakage and environmental impacts are the most concerned problems for risk assessments of CO2 storage, which are closely related to fluid (gas) transportation (diffusion and permeation) in clay mineral.
4. Conclusions
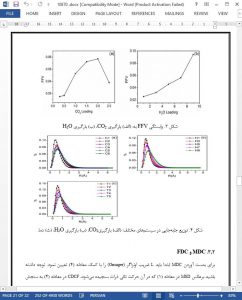
CO2 diffusion and permeation in Mt under varying conditions is investigated by MD. The results show that while both water content and temperature are positively correlated to the SDC, the SDCs of CO2 and H2O display a peak with the increase of CO2 concentration. To explain the unusual findings of the SDC further, FFV within Mt and the displacement distribution of CO2 is analyzed. It is found that FFV has an important effect on the diffusion of gas molecules in Mt. The increase of CO2 concentration causes the Mt to expand, increasing the internal FFV of Mt and providing gas molecules with more space for diffusion. This explains the initial increase of the fluid SDC in Mt. FFV begins to decrease when the CO2 concentration rises above 2 molecules/unit cell, which hinders the gas diffusion and therefore leads to decreasing SDC. Similarly, the increase of water content also causes the Mt to expand and creates more free space for CO2 diffusion, resulting in increasing CO2 SDC. It can be drawn that SDC of the fluid in Mt depends on the FFV of Mt. Furthermore, the displacement distribution of CO2 follows log-normal distribution, and the mean of the distribution shows the same trend as the SDC, which provides a good explanation of the effects of CO2 concentration, moisture and temperature on SDC from another perspective.