Abstract
Functional magnetic resonance imaging (fMRI) serves as a critical tool for presurgical mapping of eloquent cortex and changes in neurological function in patients diagnosed with brain tumors. However, the blood-oxygen-level-dependent (BOLD) contrast mechanism underlying fMRI assumes that neurovascular coupling remains intact during brain tumor progression, and that measured changes in cerebral blood flow (CBF) are correlated with neuronal function. Recent preclinical and clinical studies have demonstrated that even low-grade brain tumors can exhibit neurovascular uncoupling (NVU), which can confound interpretation of fMRI data. Therefore, to avoid neurosurgical complications, it is crucial to understand the biophysical basis of NVU and its impact on fMRI. Here we review the physiology of the neurovascular unit, how it is remodeled, and functionally altered by brain cancer cells. We first discuss the latest findings about the components of the neurovascular unit. Next, we synthesize results from preclinical and clinical studies to illustrate how brain tumor induced NVU affects fMRI data interpretation. We examine advances in functional imaging methods that permit the clinical evaluation of brain tumors with NVU. Finally, we discuss how the suppression of anomalous tumor blood vessel formation with antiangiogenic therapies can ‘‘normalize’’ the brain tumor vasculature, and potentially restore neurovascular coupling.
Neurovascular coupling is the relationship between neural firing and concomitant changes in cerebral blood flow to accommodate changing energy demands.1,2 This relationship constitutes the basis of the blood-oxygen-level-dependent (BOLD) contrast mechanism that underlies functional magnetic resonance imaging (fMRI) of the brain. However, the BOLD signal is not perfectly correlated with neuronal action potentials. The BOLD signal is a mixture of several phenomena including changes in cerebral blood flow (CBF), cerebral blood volume (CBV), and the cerebral metabolic rate for oxygen consumption (CMRO2).3 Nonetheless, it is this neurovascular coupling that has made fMRI the workhorse for interrogating and mapping brain function in patients.4
Conclusion
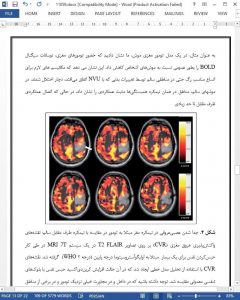
Gliomas result in varying degrees of perturbation to the neurovascular unit composed of astrocytes, pericytes, and endothelial cells. These disruptions impair neurovascular coupling and can lead to faster excitotoxicity due to the lack of vascular response and an inability to supply metabolites and oxygen to active neuronal populations. The resulting hypoxia in conjunction with tumor cell death eventually triggers a cascade of events that leads to angiogenic growth and poorly formed vasculature to support tumor growth. A shift to the co-optive phenotype, in which glioma cells take over control of host vascular tone, can also occur in patients.